Leave Your Message
-
Phone
-
E-mail
-
Whatsapp
-
Wechat


In the realm of pharmaceutical manufacturing, safety and compliance are paramount. Dr. John Stevens, a renowned expert in the field, aptly states, "A Glass Isolator is not just a tool; it’s a shield for our products." This highlights the critical role that Glass Isolators play in maintaining product integrity and protecting against contamination.
Glass Isolators create sterile environments essential for sensitive processes. They facilitate the handling of sterile materials, ensuring that no harmful particles disrupt the manufacturing process. The ability to integrate advanced technologies, such as real-time monitoring systems, further enhances their effectiveness. However, some manufacturers overlook the importance of regular maintenance. Neglect can lead to compromised environments and affect product quality.
Additionally, while Glass Isolators are vital, their installation and operation require thorough training. Insufficient training can result in errors. Achieving the ideal balance between automation and human oversight is challenging but necessary. As the pharmaceutical industry evolves, the demand for robust solutions like Glass Isolators will continue to grow, driving innovation and safety standards higher.

In pharmaceutical manufacturing, controlling contamination is paramount. Glass isolators provide a critical barrier that helps maintain sterile conditions. A recent report from the International Society for Pharmaceutical Engineering (ISPE) indicates that over 80% of contamination issues arise from human interaction. By utilizing glass isolators, companies can significantly reduce this risk.
These isolators create a controlled environment. They minimize the chances of airborne particles and microbial contamination. According to the FDA, facilities that implement isolator technology report a 30% decrease in contamination incidents. This data highlights the importance of glass isolators in protecting product integrity. The isolators allow for safe operation without compromising sterility in processes like aseptic filling and compounding.
However, it’s essential to recognize limitations. Regular maintenance and training for personnel are crucial. Many facilities overlook proper training, which can lead to breaches in sterility. As manufacturing processes evolve, so must the strategies to ensure quality and compliance. Continuous improvement in isolator technology and usage is necessary to address ongoing challenges in contamination control.
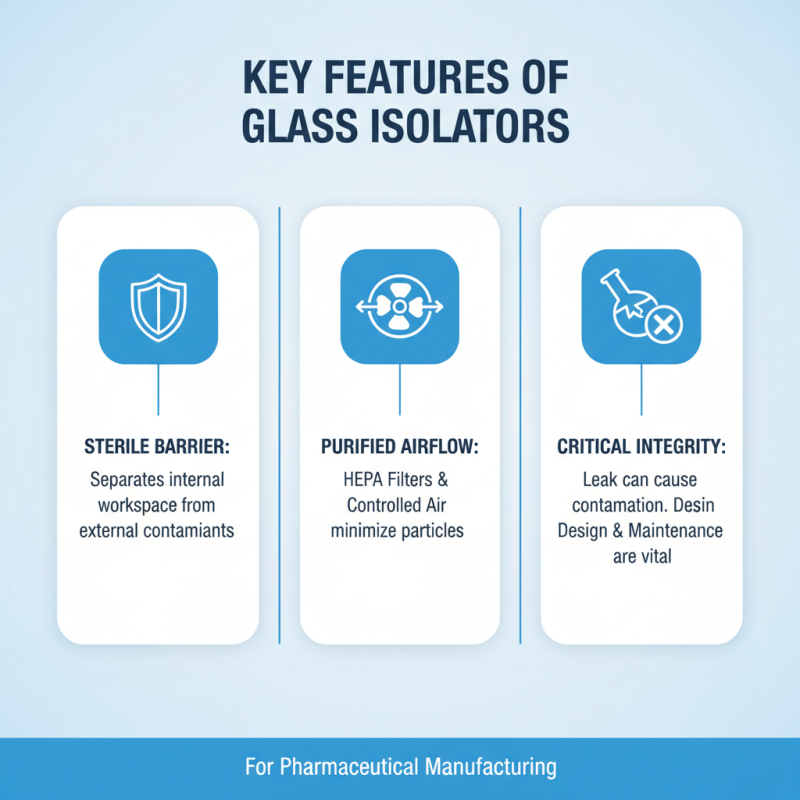
Glass isolators are critical for maintaining a sterile environment in pharmaceutical manufacturing. They provide a barrier between the internal workspace and external contaminants. Their design includes HEPA filters and controlled airflow, which minimize the risk of airborne particles. A simple leak can undermine the entire process, leading to contamination. This is a significant area for reflection in design and maintenance.
Another feature of glass isolators is their easy visibility. Operators can monitor processes without opening the unit. This reduces the chance of contamination. However, frequent checking can lead to distractions. Ensuring operators remain focused is vital. Human error, even in sterile environments, can lead to serious consequences. Regular training and reminders can help mitigate such risks.
Moreover, glass isolators can sometimes limit space. The compartments may feel cramped when handling large equipment. Finding a balance between compact design and functionality is crucial. Some manufacturers may overlook this aspect, resulting in operational inefficiencies. Constant evaluation of the layout and processes can lead to improvements in sterile production.
The pharmaceutical industry faces strict regulations. Glass isolators play a key role in meeting these standards. They ensure a controlled environment for drug production. According to a report by the International Society for Pharmaceutical Engineering (ISPE), over 80% of sterile drug manufacturing processes require isolation technology.
Glass isolators help reduce contamination risks. They provide an effective barrier against airborne particles. The FDA emphasizes the need for stringent contamination control in its sterility assurance guidelines. Reports indicate that facilities using glass isolators have seen contamination rates drop by nearly 50%. This significant reduction highlights the importance of compliance.
Despite their benefits, challenges remain. Some manufacturers struggle with integration costs and technical complexity. Employees need extensive training to operate these systems effectively. A survey revealed that 30% of organizations experienced difficulties in staff training for isolator operations. These hurdles remind us that while glass isolators are essential, implementing them requires careful planning and resources.
Glass isolators are crucial in pharmaceutical manufacturing. They provide a controlled environment that minimizes contamination risks. Their design allows for safe handling of potent compounds, protecting both the product and the operators. This level of isolation is hard to achieve with many other containment systems.
When comparing glass isolators to conventional laminar flow hoods, the differences are stark. Laminar flow hoods only provide partial protection. They direct airflow to shield products, but they can still allow airborne contaminants. Glass isolators, on the other hand, ensure a completely enclosed space. This makes them superior in maintaining product integrity.
Tip: Regular maintenance of glass isolators is essential. Proper cleaning and inspection protocols can help ensure optimal performance.
Another option is the use of RABS (Restricted Access Barrier Systems). While they offer some benefits, they don't match the full isolation of glass systems. RABS requires operators to work outside the barrier, increasing risks.
Tip: Consider the specific needs of your operation. Choosing the right system can make a huge difference. Not every facility will benefit from the same solution.
The pharmaceutical industry is evolving rapidly. Innovations in
glass isolator technology are crucial for
maintaining sterile environments. These systems protect products from contamination, ensuring
high-quality output. However, challenges persist. Some
facilities struggle with integrating advanced technology smoothly.
Recent advancements focus on enhancing efficiency. Smart sensors are now
being used. They monitor environmental conditions in real-time, providing critical data for operators.
These sensors help prevent potential breaches. Yet, there can be issues with false alarms,
leading to unnecessary downtime. Operators must constantly assess these systems.
Another area of innovation is automation. Automated glass isolators aim
to reduce human interaction. This minimizes the risk of contamination. However, the reliance on technology
raises questions. What happens if the system fails? Regular maintenance is necessary, but it can be a
logistical challenge. Flexibility in operations is
essential as manufacturers adapt to these new technologies.






